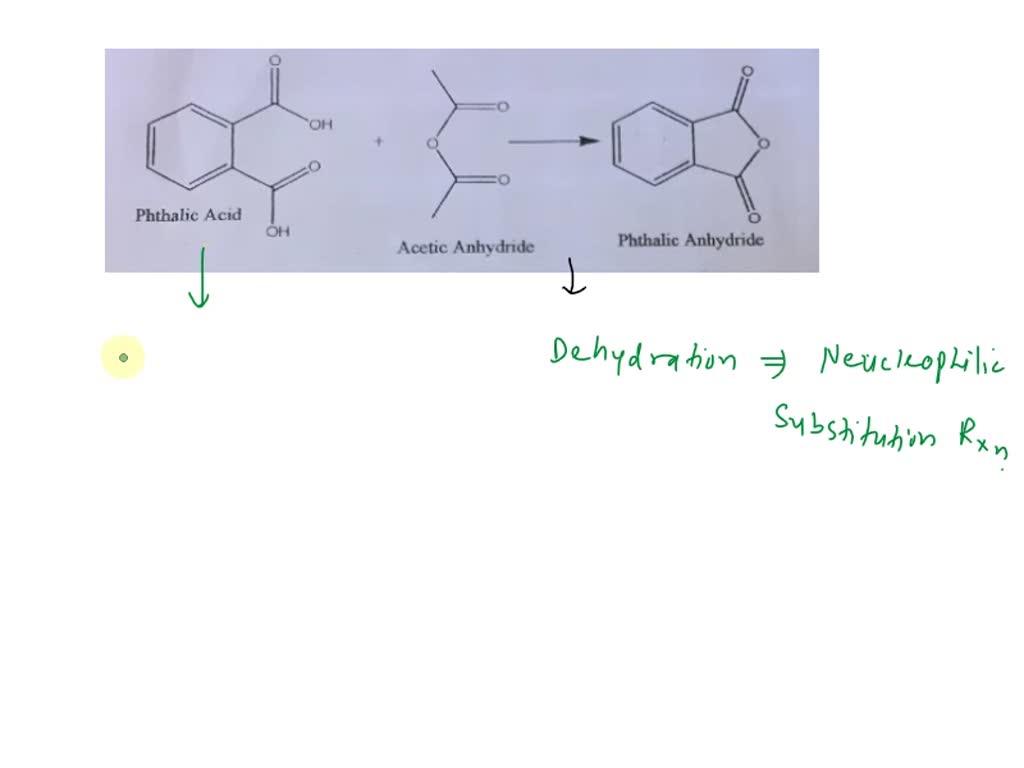
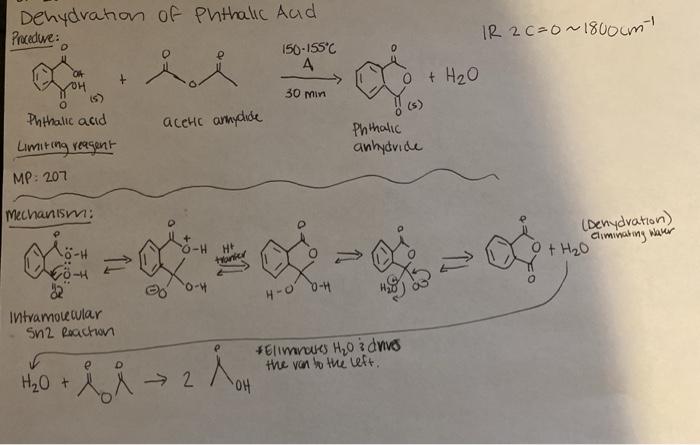
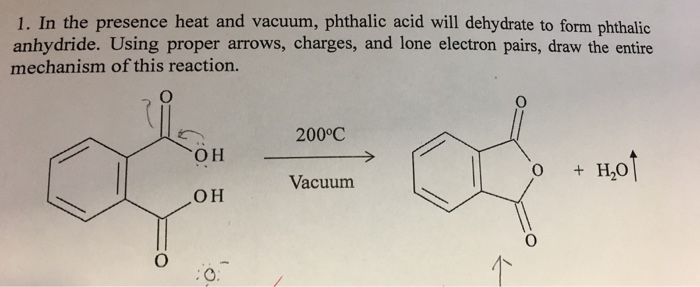
organic chemistry - Why is there no such substance as p-phthalic anhydride - Chemistry Stack Exchange
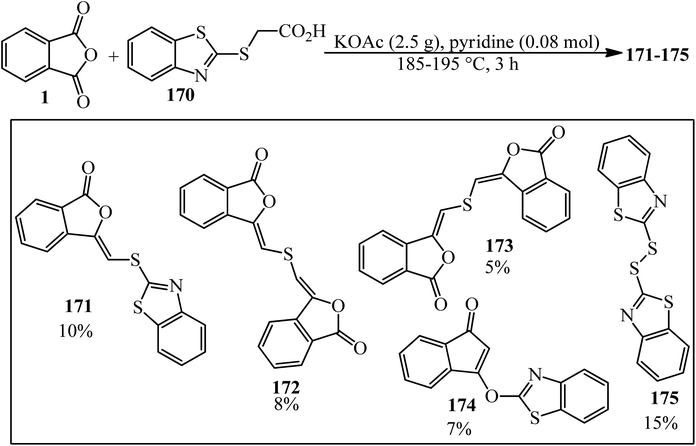
Phthalic anhydride (PA): a valuable substrate in organic transformations - RSC Advances (RSC Publishing) DOI:10.1039/D3RA03378C

Propose an arrow-pushing mechanism for the reaction between phthalic anhydride, nitric acid, and sulfuric acid to produce 3-nitrophthalic acid. | Homework.Study.com
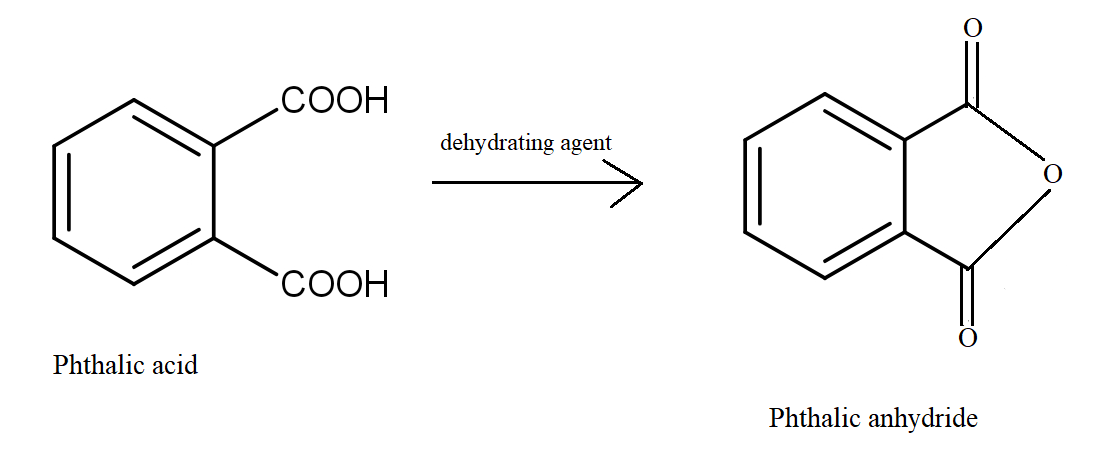
Which dicarboxylic acid in presence of a dehydrating agent is least reactive to give an anhydride?A.\n \n \n \n \n B.\n \n \n \n \n C.\n \n \n \n \n D.\n \n \
Renewable production of phthalic anhydride from biomass-derived furan and maleic anhydride - Green Chemistry (RSC Publishing) DOI:10.1039/C3GC41655K
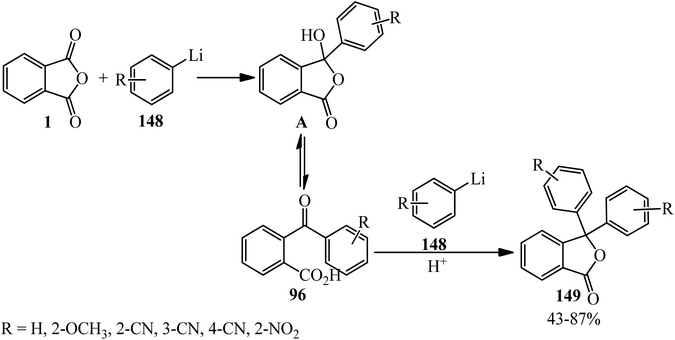
Phthalic anhydride (PA): a valuable substrate in organic transformations - RSC Advances (RSC Publishing) DOI:10.1039/D3RA03378C
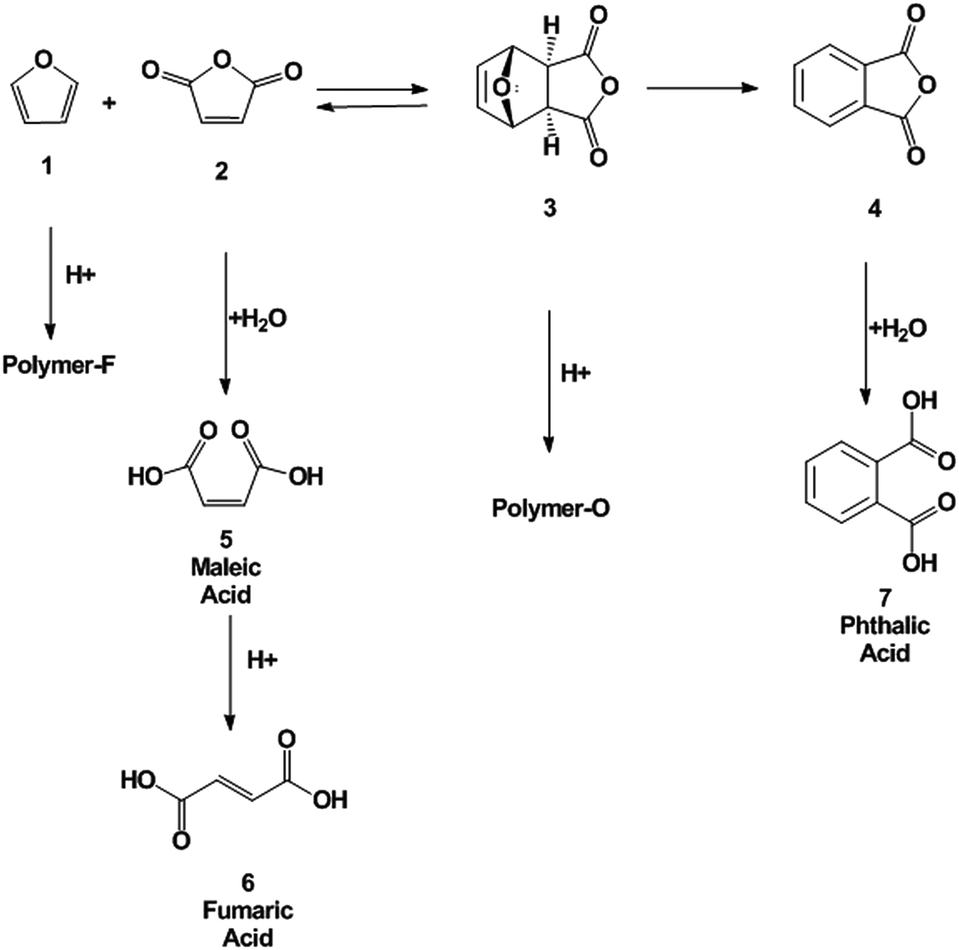
Renewable production of phthalic anhydride from biomass-derived furan and maleic anhydride - Green Chemistry (RSC Publishing) DOI:10.1039/C3GC41655K
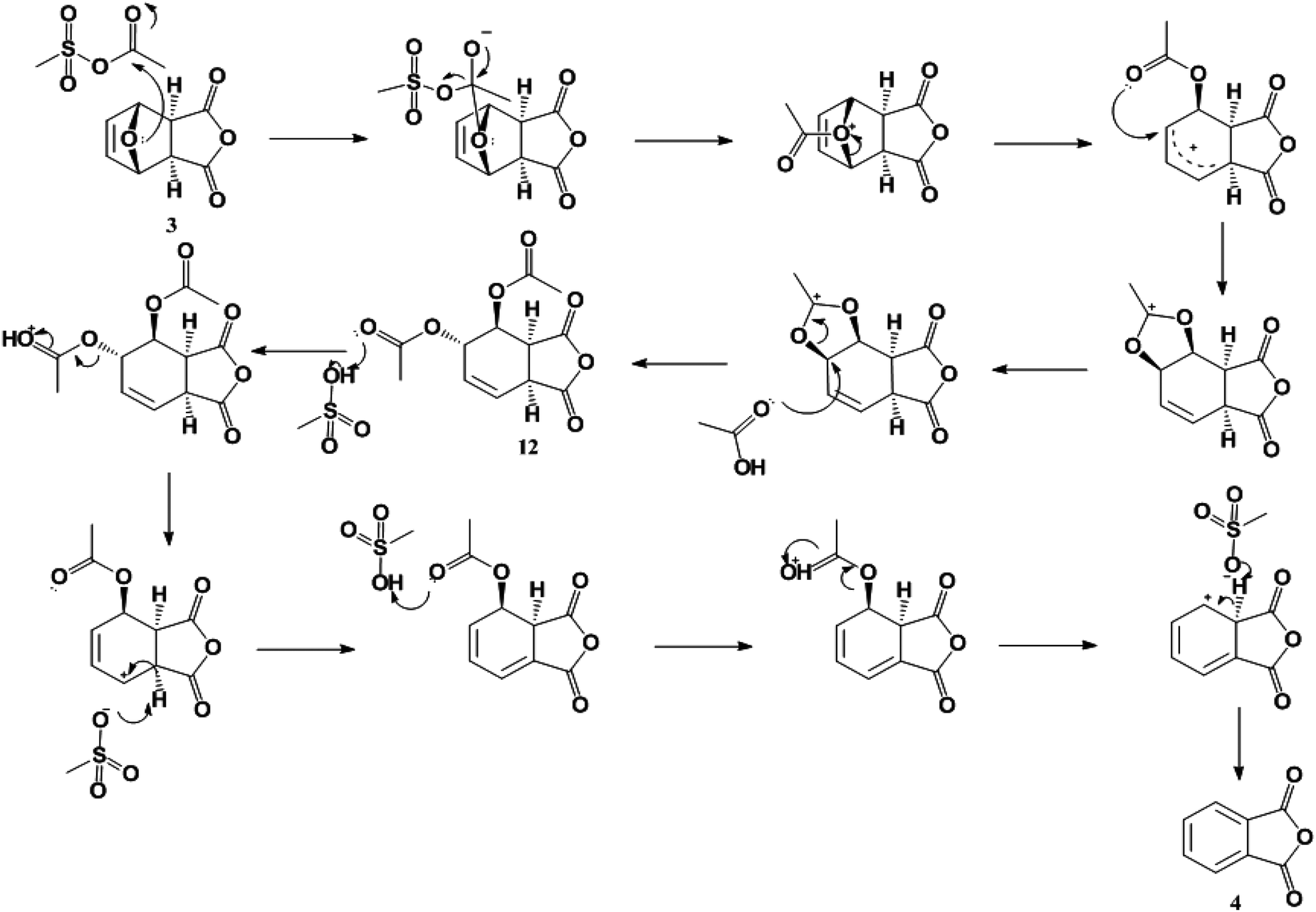
Renewable production of phthalic anhydride from biomass-derived furan and maleic anhydride - Green Chemistry (RSC Publishing) DOI:10.1039/C3GC41655K